Soil pH and Lime: How Lime Affects your Soil

What is pH?
Soil pH is the measure of its acidity or alkalinity and is rated on a simple logarithmic scale. The scale represents hydrogen ion concentration and ranges from 0.0-14.0, with 0 being the most acidic and 14 being the most alkaline. The halfway point on the scale, 7.0, is neutral. Soil acidity increases as values decrease from 7.0 to 0.0, and soil alkalinity increases as pH values increase from 7.0-14.
What causes the pH of the soil to be acidic?
The soils of the northeastern region of the United States tend to be naturally acidic. The amount of annual rainfall largely determines whether soils will become acidic, neutral, or alkaline. In regions of high rainfall, such as here in New Jersey, the alkaline elements are leached deep into the lower soil regions by percolating rain water. This natural process leads to acidic soils.
Along with rainfall, there are other factors that will affect soil pH. Removal of grass clippings that contain alkaline elements, instead of allowing them to decompose into soil, will promote acidity. Leaves, pine needles, and other plant matter can create more acidic soil conditions as they decompose, and living plants will feed on alkaline elements (potassium, calcium, and magnesium). For these reasons, areas under dense tree cover or anywhere grass and tree roots compete for soil nutrients tend to have more acidic soil.
Why does the soil pH matter?
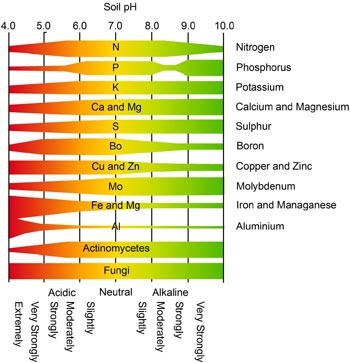 Knowing the soil pH is crucial because it strongly effects grass growth. Soil pH dictates nutrient availability, elemental toxicity, and microbial activity.
Knowing the soil pH is crucial because it strongly effects grass growth. Soil pH dictates nutrient availability, elemental toxicity, and microbial activity.
Various mineral nutrients are readily available in varying concentrations depending on the pH of the soil. At certain critical levels, some of the minerals remain bound to other minerals and are unavailable for plant use. The chart is a general representation of plant nutrient availability based on soil pH levels. The narrow areas of each band represent low availability of that nutrient, while the taller areas represent optimal nutrient availability.
The chart clearly illustrates that between a pH of 6.0-7.0, availability is at its peak for most of the critical lawn nutrients. The other thing to observe is that at about 5.5, nutrient availability becomes problematic and only gets worse as the soil becomes more acidic. Likewise, as the soil becomes more alkaline than 7.0, nutrient availability will also begin to suffer. However, soils with a pH of 7.0 or more are very rare in New Jersey, so the concern is normally in keeping lawn soil as alkaline as possible. The chart is not specific to our grass types, for typical cool season grasses found in New Jersey, the recommend pH level for optimal nutrient availability is between 6.3 and 6.5.
In addition to increased soil nutrient availability at a range of 6.0-7.0, this is also the range at which microorganism activity starts to peak. On the above chart the line labeled actinomycetes illustrates this point. Actinomycetes are bacteria in the soil responsible for the breakdown of a lot of organic matter as well as complex soil nutrients. Keeping the soil microorganisms as active as possible is of interest to the lawn care technician because they will help breakdown fertilizers into forms usable by plants, as well as keep thatch to a minimum by aiding in decomposition.
How can the pH be corrected?
Acidic soil pH can be corrected by applying lime. The most common liming materials are calcitic or dolomitic agricultural limestone. These are natural products made by finely grinding natural limestone. Since natural limestone is relatively water insoluble, agricultural limestone must be very finely ground so that it can mix with the soil particles and react with other nutrients to change soil acidity. The more finely ground the limestone is, the faster it will react in the soil. Both calcitic, and dolomitic lime contain calcium carbonate. Dolomitic lime however, also contains magnesium in the form of magnesium carbonate and should be used when soil tests indicate a magnesium deficiency.
Because high quality, finely ground limestone is very dusty and difficult to spread, some companies market a prilled or pelletized limestone for commercial and residential use. A small amount of clay or a polymer is added to the ground limestone so small prills are formed instead of dust. This makes it easier to apply out of a fertilizer spreader. Once applied, soil moisture will cause the granules to dissociate and disperse the limestone particles. Furthermore, newer pelletized lime products can be manufactured with reactants such as organic acids. These reactants speed up the chemical process by which lime changes soil pH, allowing for lawn applicators to produce quicker results, while using less product.
When using any limestone product, it is important to apply the material at the correct rate. Calcitic or dolomitic limestone, be it ground or pelletized, can have recommended application rates ranging anywhere from 5-200lbs/1000sq.ft. The rate at which the lime should be applied depends on the pH of the soil, what target range is trying to be obtained, and soil type.
 Optimum pH range in cool-season turf soil is between 6.3-6.5. At this range soil microbe activity and nutrient availability is high, and it provides the most optimum condition for the most desirable species of Kentucky bluegrass and perennial rye.
Optimum pH range in cool-season turf soil is between 6.3-6.5. At this range soil microbe activity and nutrient availability is high, and it provides the most optimum condition for the most desirable species of Kentucky bluegrass and perennial rye.
Soil type comes into play when determining lime application rate. Certain soil types will be more resistant to change in pH than others. In general, it is more difficult to change pH in clay soil than it is in sandy soil. When liming clay, higher lime amounts will be necessary to have the same effect that a smaller amount would have on sandy soils.
Before applying lime to an area, a test of the pH should be done. Soil pH should be measured at least once a year as a general practice. This can be done using any number of kits or through the use of a pH meter. The lower the pH, the more lime it will take to correct. For example, a soil with a pH of 4.5 will require significantly more lime to correct to 6.5 than a lawn with an initial pH of 6.0. It is for this reason that a pH reading should be taken at least once each year.
If severely low pH is suspected, a soil sample can be sent to a commercial or university lab. The results of the soil test will specify how much lime should be added in a single or multiple applications to correct the problem. In addition, any other nutrient deficiencies that may exist will be specified, and recommendations on treatment to correct these issues will be provided. Soil tests are a very accurate and a useful tool in determining hard to diagnose turf issues. However, soil test results take time and are an added expense, which is why they are less practical for determining pH on every lawn.
If you are in our service area and want to know more about liming your lawn or testing the pH, give our office a call at 908-281-7888. Also, you can learn more about our liming process from our website.




